Group vom Berg
Local immunomodulation for the treatment of brain cancer
Immune responses are tightly regulated by a system of soluble and membrane bound mediators to exchange information between cellular actors in a spatially and temporally controlled manner. Most of these signaling molecules are used to transmit information upon direct cell-to-cell contact or over distances ranging from a few cell diameters to anatomically confined organ systems. Yet, despite these well-known facts systemic, intravenous (i.v.) application has often been favored over local administration in cancer immunotherapy, where cytokines or checkpoint blockers manipulate such signaling to raise a tumor clearing immune response. This means, unfortunately, that for effective treatment responses this approach leads to high whole-body exposure which in turn increases the risk for treatment related adverse events (trAE).
Glioblastomas (GB) arise in the central nervous system (CNS), a well confined anatomical structure. GB constitutes a usually unifocal cancer entity. GB therefore represents an ideal target for local immunotherapy. We could show, however, that when the aim of local application is to avoid trAE the simple approach of delivering immune-checkpoint blocking antibodies or cytokines intratumorally instead of systemically will only marginally increase the harm-to-benefit ratio.
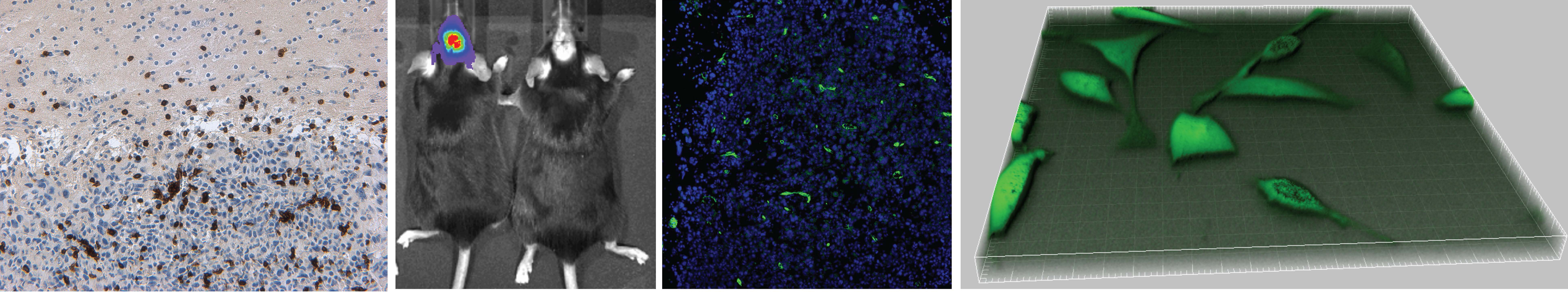
Improved biologicals for local immunotherapy of neurological diseases
One of our goals is to adapt antibody-based biologicals for high local tissue retention upon local delivery to have high concentration in the brain tumor and low concentration in the blood. As antibodies are useful to treat neurological diseases beyond brain cancer, we are also studying local delivery into the brain parenchyma, not just into the brain tumor. For the mechanical delivery, we use convection-enhanced delivery (CED) a method that has already been used for treating GB and Parkinson’s Disease (PD).
One health – comparative oncology in dogs and humans
Humans “best friend” shares not only most of the living circumstances (“exposome”) with its owner but also many of the diseases. Depending on the breed, some are highly prone to diabetes, arthritis or cancer. Compared to humans, the arsenal to treat companion dogs with cancer is very small and lagging behind decades. In collaboration with veterinarians we study the proportion and functional state of immune cells in the blood of companion dogs that present at the hospital with cancer. In cell culture, we screen drugs used for humans to check whether they also work for dogs to eventually use them to treat not only humans but also dogs with cancer.
